Atomic structure of fluorine11/27/2023 
The 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but, as they are found in different energy levels, they occupy different spaces around the nucleus. 
Orbitals on different energy levels are similar to each other, but they occupy different areas in space. The energy level is determined by the period and the number of electrons is given by the atomic number of the element. The p, d, and f orbitals have different sublevels, thus can hold more electrons.Īs stated, the electron configuration of each element is unique to its position on the periodic table. The four different types of orbitals (s,p,d, and f) have different shapes, and one orbital can hold a maximum of two electrons. Electrons exhibit a negative charge and are found around the nucleus of the atom in electron orbitals, defined as the volume of space in which the electron can be found within 95% probability. Every element on the periodic table consists of atoms, which are composed of protons, neutrons, and electrons. The valence electrons, electrons in the outermost shell, are the determining factor for the unique chemistry of the element.īefore assigning the electrons of an atom into orbitals, one must become familiar with the basic concepts of electron configurations. 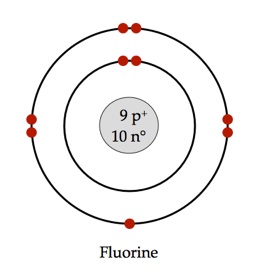
Many of the physical and chemical properties of elements can be correlated to their unique electron configurations. The polymer is inert to most chemicals, does not melt below 327 C (620 F), and has the lowest coefficient of friction of any known solid. Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain of electrons in their subsequent orbitals. The fluorine atoms surround the carbon chain like a protective sheath, creating a chemically inert and relatively dense molecule with very strong carbon-fluorine bonds. The electron configuration of an atom is the representation of the arrangement of electrons distributed among the orbital shells and subshells.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
